Balanced Chemical Equation ↓
C2H5OH + 3O2 → 2CO2 + 3H2OThe gas laws played a big part in this project.
For example, we made a small gas chamber instead of a big one, because Boyle's Law states that, the volume of the gas is inversely proportional to its pressure. We then estimated that the smaller the chamber is, the more pressure is unleashed when it is fired. Charle's Law states that, volume and temperature are directly proportional. So, the smaller the volume the higher the pressure as well. The key to this part is that the chamber cannot be so big that the pressure won't build, but not so small that the bottom of the can doesnt blow up.
 Gay-Lussacs Law
Gay-Lussacs Law Charles Law
Charles Law Boyle's Law
Boyle's Law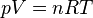 Ideal Gas Law
Ideal Gas LawThis also surprisingly involves Gay-Lussac's Law, Gay-Lussac's Law states that the pressure and temperature are directly proportional. The problem this poses is that if the temperature is cold outside like it is now and will be for a few more weeks, then the pressure will not build up as we hoped it would.
No comments:
Post a Comment